Elements, Compounds and Mixtures
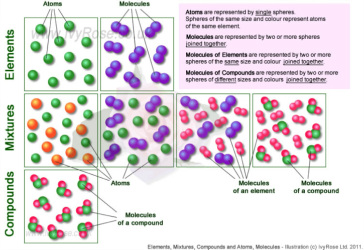
Elements – simplest form of pure substance. They cannot be broken into anything else by physical or chemical means.
Example: Lithium or Carbon
Compounds – pure substances that are of two or more elements. They can be broken into simpler substances by chemical means.
Example: H2O/Water
Mixtures – two or more substances that are not chemically combined with each other and can be separated by physical means. The substances in a mixture retain their individual properties.
Example: Salad
Solutions – a special kind of mixture where one substance dissolves in another.
Example: Hot chocolate
Example: Lithium or Carbon
Compounds – pure substances that are of two or more elements. They can be broken into simpler substances by chemical means.
Example: H2O/Water
Mixtures – two or more substances that are not chemically combined with each other and can be separated by physical means. The substances in a mixture retain their individual properties.
Example: Salad
Solutions – a special kind of mixture where one substance dissolves in another.
Example: Hot chocolate

