Phase Change
Solids
Particles of solids are tightly packed, vibrating about a fixed position.
Solids have a definite shape and a definite volume.
Liquids
Particles of liquids are tightly packed, but are far enough apart to slide over one another.
Liquids have an indefinite shape and a definite volume.
Gases
Particles of gases are very far apart and move freely.
Gases have an indefinite shape and an indefinite volume.
Plasma
A plasma is an ionized gas. That means that a substance had so much energy that the electrons were ripped off of their atoms.
Particles of solids are tightly packed, vibrating about a fixed position.
Solids have a definite shape and a definite volume.
Liquids
Particles of liquids are tightly packed, but are far enough apart to slide over one another.
Liquids have an indefinite shape and a definite volume.
Gases
Particles of gases are very far apart and move freely.
Gases have an indefinite shape and an indefinite volume.
Plasma
A plasma is an ionized gas. That means that a substance had so much energy that the electrons were ripped off of their atoms.
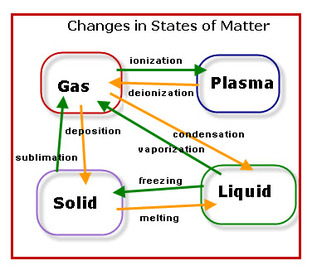
Melting: Change in state from solid to a liquid
Freezing: Change in state from a liquid to a solid
Vaporization: Change in state from liquid to gas
Condensation: Change in state from gas to liquid
Sublimation: Change in state from solid to gas
Deposition: Change in state from gas to solid
Freezing: Change in state from a liquid to a solid
Vaporization: Change in state from liquid to gas
Condensation: Change in state from gas to liquid
Sublimation: Change in state from solid to gas
Deposition: Change in state from gas to solid
Phase Change Graph

This graph shows a substance being heated at a constant rate. You'll notice that there are certain places on the graph that are flat lines meaning that the temperature is NOT increasing. Where does the energy go?
The energy goes into the phase change. It is pushing the molecules further apart so that they can begin to slide over each other like in a liquid or bounce around each other like a gas.
The lines that are diagonal indicate that the substance is in only one state and that the temperature is increasing.
Remember that the melting point is the same as the freezing point and occurs at 0 degrees Celsius for water. The boiling point is the same as the condensation point which is the same as the vaporization point. It occurs at 100 degrees Celsius for water.
The energy goes into the phase change. It is pushing the molecules further apart so that they can begin to slide over each other like in a liquid or bounce around each other like a gas.
The lines that are diagonal indicate that the substance is in only one state and that the temperature is increasing.
Remember that the melting point is the same as the freezing point and occurs at 0 degrees Celsius for water. The boiling point is the same as the condensation point which is the same as the vaporization point. It occurs at 100 degrees Celsius for water.
