Rates of Reactions

Definition: The rate of a reaction is how fast a reaction takes place.
The rate of reaction depends on the collisions between molecules. If molecules have enough energy and the correct orientation, they'll be able to collide and react.
The rate of reaction can be affected by 5 things:
The rate of reaction depends on the collisions between molecules. If molecules have enough energy and the correct orientation, they'll be able to collide and react.
The rate of reaction can be affected by 5 things:
- Temperature
- Surface Area
- Concentration
- Pressure
- Catalysts & Inhibitors
Temperature

If something is cold, the molecules aren't moving very quickly. Heat it up, and now the molecules are moving quickly. Because the molecules are moving quickly, they'll collide into other molecules more often which will increase the rate of reaction.
Surface Area
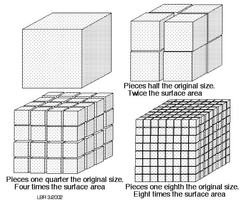
If the particles are going fast enough to react, the next step is to allow it to react with as much material as it can. That's where surface area comes in. Surface area is all about exposing as much material as possible. Look at the picture to the right. The top left cube is protecting the inside from reacting with anything. But if we cut that cube up then the inside can react at the same time as well. By cutting it up, we've increased the amount of molecules that can react at one time. More surface area means more molecules to collide with so the reaction will go faster.
Concentration
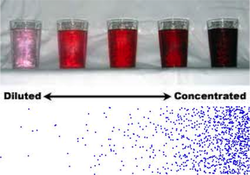
Concentration is how many particles are in a place. A diluted substances means there are not many molecules in one place. A concentrated substance is one that has lots of molecules in one place. If there are more molecules in a place, they'll be more likely to collide and react faster.
Pressure
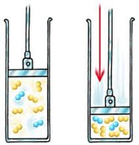
Increasing pressure decreases the space allowed for the same amount of molecules. With less space to move around in, particles will collide more often.
Think about trying to squeeze a balloon. You are decreasing the space allowed for the air in the balloon. Those molecules have to keep moving at the same speed so because they're in a smaller container, they'll hit the walls of the balloon more often and it will feel like the air is pushing back on you. Because it is!
Think about trying to squeeze a balloon. You are decreasing the space allowed for the air in the balloon. Those molecules have to keep moving at the same speed so because they're in a smaller container, they'll hit the walls of the balloon more often and it will feel like the air is pushing back on you. Because it is!
Catalyst
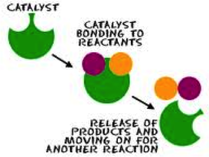
A catalyst is a substance that speeds up a reaction.
An example of a catalyst is an enzyme. Enzymes in our stomaches help us digest food faster. If we didn't have enzymes in our stomaches, food would stay in our stomaches for years!
An example of a catalyst is an enzyme. Enzymes in our stomaches help us digest food faster. If we didn't have enzymes in our stomaches, food would stay in our stomaches for years!
Inhibitor
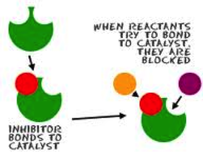
An inhibitor is a substance that slows down a reaction.
It blocks certain molecules from colliding with others.
An example of an inhibitor is food preservatives. They block oxygen from getting to the food and rotting it.
It blocks certain molecules from colliding with others.
An example of an inhibitor is food preservatives. They block oxygen from getting to the food and rotting it.
Watch this youtube video. It will help you understand the 5 rates of reaction and it will give you a laugh too!
