Atoms
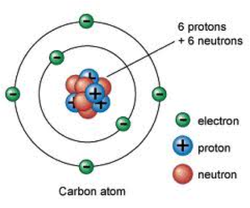
•Atom – The smallest component of an element consisting of a nucleus of protons and neutrons with electrons circling this nucleus.
Elements

•Element – A substance composed of atoms that are all the same.
Example: diamond (all carbon atoms) and graphite (also all carbon atoms)
Example: diamond (all carbon atoms) and graphite (also all carbon atoms)
The Periodic Table

It's easy to find the number of protons and electrons in an atom because it's just the top number or the Atomic Number. The number of neutrons is found by subtracting the atomic number from the atomic mass.
Atomic Mass - Atomic Number = Neutrons
23 - 11 = 12
Atomic Mass - Atomic Number = Neutrons
23 - 11 = 12
